|
7/31/2023 0 Comments H molar mass 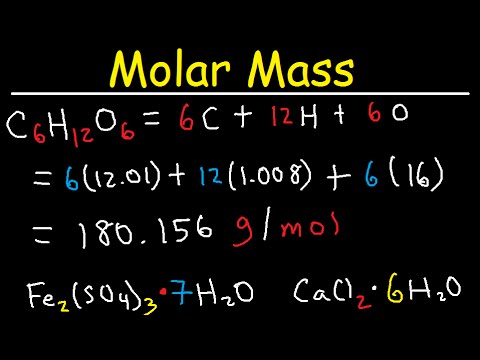
Since hydrogen does not exist as one atom, the molar mass of the hydrogen molecule is 2.016 g/mol. 
There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. The molar mass of an element or molecule is the total mass in grams of all the atoms that comprise a mole of a certain molecule. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. It is also sometimes called: Molecular Mass, Molecular Weight, Formula Mass, or Formula Weight. 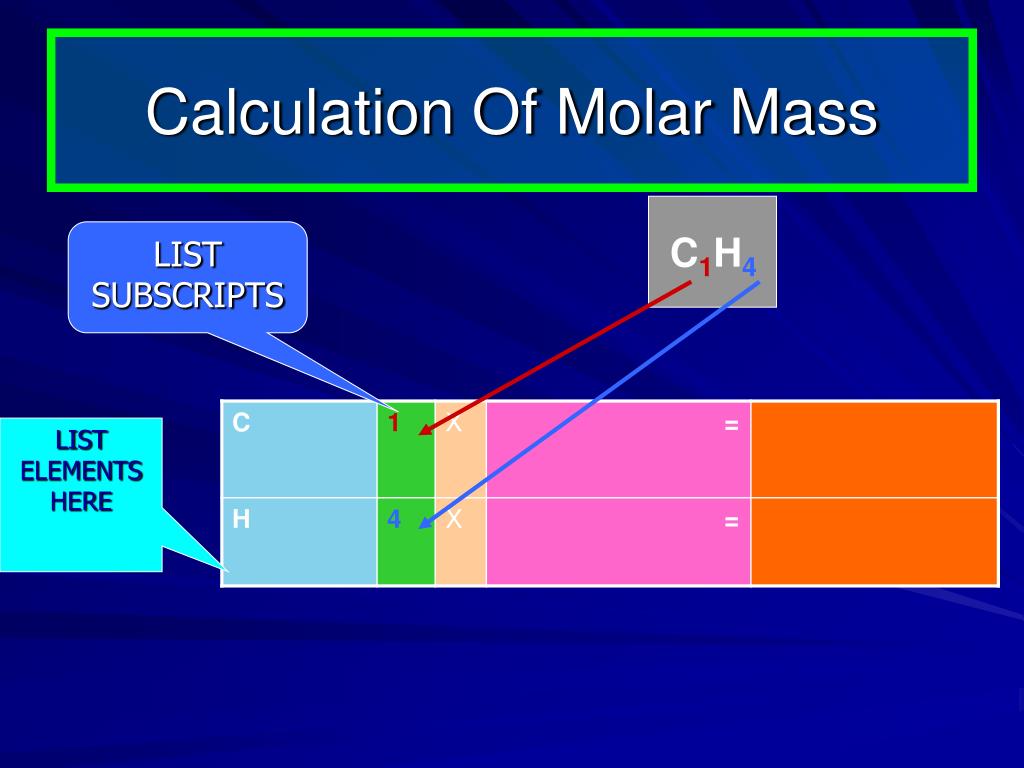
The chemical identity of a substance is defined by the types and relative numbers of atoms composing its fundamental entities (molecules in the case of covalent compounds, ions in the case of ionic compounds). One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. The mass in grams of 1 mole of substance is its molar mass. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Molar mass is the mass (in atomic mass units) of one mole of a of a substance. The molar mass of any substance is the mass in grams of one mole of representative particles of that substance. This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. Molar Mass of an Element According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Plant Inspection & Process Optimalisation Now, the molar mass of the molecule can be calculated according to the number of atoms present in the molecule. As an example, the molecular weight of water, H2O, is calculated as (2 × 1.00797) + (1 × 15.9994) 18.0153, where 1.00797 and 15.9994 are the atomic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
